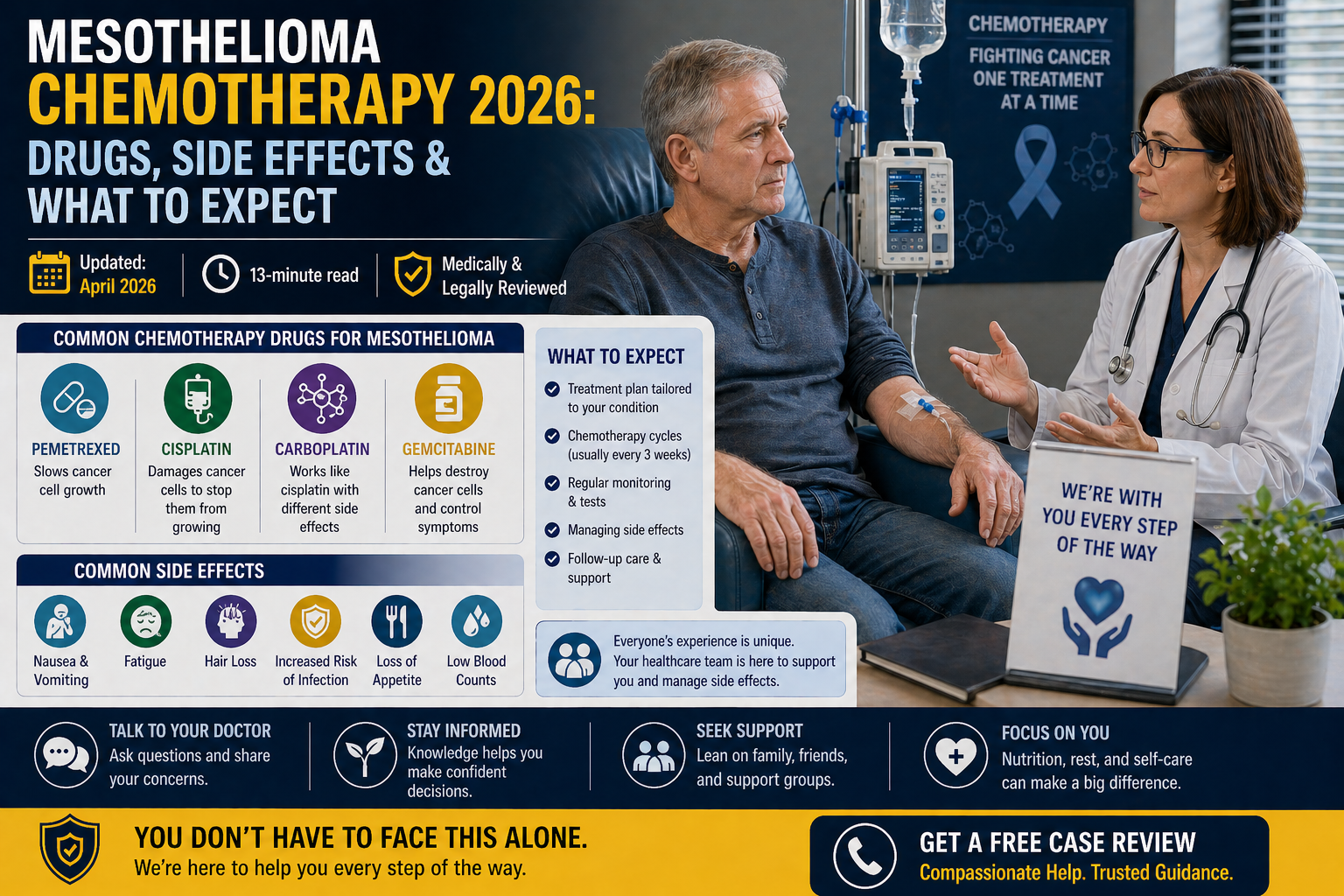
📅 Updated: April 2026 | ⏱ 13-minute read | ✅ Medically & Legally Reviewed
Introduction
Chemotherapy has been the backbone of mesothelioma treatment for over two decades. While immunotherapy has transformed outcomes for many patients since 2020, chemotherapy remains a cornerstone of care, used as a first-line treatment, before surgery, after surgery, and in combination with newer targeted therapies.
Understanding chemotherapy for mesothelioma in 2026 means understanding more than just the drugs. It means knowing why certain regimens are chosen over others, what the side effects actually feel like and how they’re managed, what survival data says about chemo’s effectiveness, and how chemotherapy treatment costs can be recovered through legal compensation.
This guide covers all of it, clearly, honestly, and with the most current 2026 clinical data available.
Why Chemotherapy Still Matters in 2026
With FDA-approved immunotherapy (nivolumab + ipilimumab) now available and clinical trials pushing toward cancer vaccines and CAR T-cell therapy, it’s natural to wonder: is chemotherapy still relevant?
The answer is yes, for several important reasons:
1. Chemotherapy remains first-line for many patients
For epithelioid mesothelioma specifically, the 2024 ASCO guideline update confirmed that chemotherapy, either alone or combined with immunotherapy, remains an appropriate and evidence-supported first-line option. Not every patient is a candidate for or chooses immunotherapy.
2. Chemotherapy is used before and after surgery
Neoadjuvant chemotherapy (before surgery) shrinks tumors to improve surgical outcomes. Adjuvant chemotherapy (after surgery) eliminates residual microscopic disease. Surgery-based treatment protocols are incomplete without chemotherapy in most cases.
3. Chemotherapy is the foundation of combination regimens
The landmark KEYNOTE-483 trial added pembrolizumab to standard pemetrexed + platinum chemotherapy, a recognition that chemotherapy’s tumor-targeting mechanism and immunotherapy’s immune activation work synergistically together.
4. Chemotherapy is accessible and covered
Unlike some experimental therapies still in trials, pemetrexed-based chemotherapy is FDA-approved, widely covered by Medicare and commercial insurance, and available at virtually every oncology center in the country.
Mesothelioma Chemotherapy Drugs, The Complete 2026 Guide
First-Line Standard: Pemetrexed + Cisplatin
The standard first-line chemotherapy regimen for mesothelioma has been pemetrexed (Alimta) combined with cisplatin since the drug combination was FDA-approved in 2004, based on the landmark Vogelzang trial that demonstrated a 12.1-month median survival versus 9.3 months with cisplatin alone.
Two decades later, it remains the most thoroughly studied and most commonly used chemotherapy regimen for mesothelioma.
How it works:
| Drug | Drug Class | Mechanism |
|---|---|---|
| Pemetrexed (Alimta) | Antifolate | Inhibits multiple folate-dependent enzymes required for DNA synthesis, starves cancer cells of the building blocks they need to divide |
| Cisplatin | Platinum compound | Cross-links DNA strands inside cancer cells, triggers cell death by making DNA replication impossible |
Together, these two drugs attack cancer cell division through complementary mechanisms, which is why the combination is more effective than either drug alone.
Treatment schedule:
- Both drugs are administered intravenously (IV) in an infusion center
- Standard cycle: Every 21 days (3 weeks)
- Typical course: 4–6 cycles (12–18 weeks of treatment)
- Pre-medication with folic acid and vitamin B12 is required before starting pemetrexed, this significantly reduces side effects without reducing effectiveness
Response rates:
| Metric | Pemetrexed + Cisplatin |
|---|---|
| Objective response rate | 41.3% |
| Disease control rate | ~70% |
| Median overall survival | 12.1 months |
| Median progression-free survival | 5.7 months |
Source: Vogelzang et al., Journal of Clinical Oncology, 2003, still the foundational reference for this regimen
First-Line Alternative: Pemetrexed + Carboplatin
For patients who cannot tolerate cisplatin, due to kidney function issues, hearing problems, neuropathy, or other comorbidities, pemetrexed + carboplatin is an equally effective alternative with a more manageable side effect profile.
Key differences from cisplatin:
| Factor | Cisplatin | Carboplatin |
|---|---|---|
| Route | IV infusion | IV infusion |
| Kidney toxicity | Higher | Lower |
| Nausea | More severe | Less severe |
| Neuropathy risk | Higher | Lower |
| Hearing loss risk | Present | Minimal |
| Efficacy vs. pemetrexed | Equivalent | Equivalent |
| Typical choice | Younger, better kidney function | Older patients, kidney issues |
Multiple studies have confirmed that pemetrexed + carboplatin achieves equivalent survival outcomes to pemetrexed + cisplatin, making it not a “lesser” option, but an equally valid one tailored to patient tolerance.
Chemoimmunotherapy: Pembrolizumab + Pemetrexed + Platinum (KEYNOTE-483)
One of the most significant chemotherapy developments of 2025 was the FDA approval of pembrolizumab (Keytruda) combined with pemetrexed and a platinum drug as a first-line treatment for unresectable pleural mesothelioma.
This approval was based on the KEYNOTE-483 Phase III trial, which directly compared this chemoimmunotherapy combination against standard chemotherapy alone.
KEYNOTE-483 Results:
| Metric | Pembro + Chemo | Chemo Alone |
|---|---|---|
| Median Overall Survival | 17.3 months | 16.1 months |
| Median PFS | 7.1 months | 7.0 months |
| Objective Response Rate | 56.7% | 48.1% |
| Grade 3–4 Adverse Events | 73.5% | 65.1% |
Source: FDA approval documentation, October 2025; KEYNOTE-483 full results
What this means for patients: The addition of pembrolizumab to standard chemotherapy modestly improved overall survival and notably improved response rates. This option is now particularly relevant for epithelioid mesothelioma patients who prefer a chemotherapy-based first-line approach with immunotherapy added, rather than immunotherapy-only (nivolumab + ipilimumab).
Bevacizumab (Avastin), Adding an Anti-Angiogenic Agent
Bevacizumab is an anti-angiogenic drug, it blocks VEGF (Vascular Endothelial Growth Factor), a protein that tumors use to grow new blood vessels to feed their expansion.
The MAPS trial (French Cooperative Thoracic Intergroup) evaluated adding bevacizumab to pemetrexed + cisplatin for pleural mesothelioma. Results showed a meaningful improvement:
- Median OS: 18.8 months (pemetrexed + cisplatin + bevacizumab) vs. 16.1 months without bevacizumab
- The NCCN guidelines include bevacizumab as an add-on option for eligible patients
Who is NOT eligible for bevacizumab:
- Patients taking anticoagulants (blood thinners)
- Patients with uncontrolled hypertension
- Patients with recent major surgery (within 4 weeks)
- Patients with hemoptysis (coughing blood) or significant bleeding history
For eligible patients without these contraindications, adding bevacizumab to standard chemotherapy represents an additional survival benefit with a well-characterized side effect profile.
Second-Line Chemotherapy, After First-Line Treatment
When first-line treatment stops working, either because the disease progresses or the patient can no longer tolerate the initial regimen, second-line chemotherapy options include:
| Drug | Notes |
|---|---|
| Gemcitabine | Commonly used second-line; given IV weekly for 2 of every 3 weeks |
| Vinorelbine | Oral or IV; well-tolerated option for patients needing flexibility |
| Pemetrexed re-treatment | For patients who initially responded and had a treatment break; can re-challenge |
| Ramucirumab + Gemcitabine | Anti-VEGFR2 antibody combined with gemcitabine; showing promise in trials |
| Nivolumab (Opdivo) | May be used as second-line immunotherapy after chemotherapy failure; off-label |
Second-line response rates are lower than first-line, typically 10–20% for single-agent chemotherapy, but meaningful disease control is achievable for many patients, and progression-free survival extends several months in responding patients.
Neoadjuvant and Adjuvant Chemotherapy, Surgery Settings
Neoadjuvant (Before Surgery):
Chemotherapy is increasingly being used before P/D or EPP to:
- Shrink tumor burden and improve completeness of surgical resection
- Test whether the tumor responds to pemetrexed-based therapy (non-responders may be shifted to a different approach)
- Treat potential micrometastatic disease before the surgical field is disturbed
Typical neoadjuvant protocol: 3–4 cycles of pemetrexed + cisplatin (or carboplatin), followed by surgical evaluation.
Adjuvant (After Surgery):
Post-surgical chemotherapy targets residual microscopic disease and reduces the risk of local recurrence. Typically 2–4 cycles of pemetrexed + cisplatin/carboplatin following surgical recovery.
How Chemotherapy Is Administered
Understanding what a chemotherapy treatment day actually looks like helps reduce anxiety and allows patients to plan.
Typical Chemotherapy Infusion Day
Before treatment:
- Blood draw to check white blood cell count, platelet count, kidney function, and liver enzymes
- Results reviewed, treatment proceeds only if blood counts are adequate
- IV line or port access prepared
During infusion:
| Step | Duration |
|---|---|
| Anti-nausea medications (ondansetron, dexamethasone) | 30 minutes |
| Hydration fluids (particularly important with cisplatin) | 1–2 hours |
| Pemetrexed infusion | 10 minutes |
| Cisplatin infusion | 2 hours |
| Post-hydration flush | 1–2 hours |
| Total time at infusion center | 4–6 hours |
Carboplatin infusions are slightly shorter; cisplatin requires more extensive hydration to protect kidney function.
After treatment:
- Oral anti-nausea medications prescribed for home use
- Written instructions on side effects to watch for
- Next appointment scheduled
Most patients drive themselves to early cycles, though having a driver for the first treatment is advisable until the patient knows how they personally respond.
Mesothelioma Chemotherapy Side Effects, The Honest Guide
Side effects vary significantly by drug, dose, individual patient health, and supportive medications used. Here is a comprehensive breakdown of what to expect, and how each effect is managed in 2026.
Pemetrexed Side Effects
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Fatigue | Very common (80%+) | Mild–Moderate | Rest planning; activity pacing; review anemia |
| Nausea/Vomiting | Common | Mild–Moderate | Pre-meds (ondansetron, aprepitant); significantly reduced with folic acid + B12 |
| Mouth sores (stomatitis) | Common | Mild | Folic acid supplementation; saltwater rinse; medicated mouthwash |
| Skin rash | Common | Mild–Moderate | Dexamethasone pre-treatment prevents most rash; topical corticosteroids if needed |
| Reduced blood counts | Common | Variable | Weekly CBC monitoring; G-CSF injections (Neupogen) if white count drops |
| Loss of appetite | Common | Mild–Moderate | Nutritional counseling; small frequent meals |
| Hair thinning | Occasional | Mild | Usually partial, not complete hair loss |
| Diarrhea | Occasional | Mild–Moderate | Anti-diarrheal medications; hydration |
Key pemetrexed protocol: Supplementing with folic acid (400–1,000 mcg daily) beginning 7 days before the first dose, and vitamin B12 (1,000 mcg injection) given 1–2 weeks before the first dose and every 9 weeks, dramatically reduces toxicity without affecting drug efficacy. This is non-negotiable and should be confirmed at every cycle.
Cisplatin Side Effects
| Side Effect | Frequency | Severity | Management |
|---|---|---|---|
| Nausea/Vomiting | Very common | Moderate–Severe | Best managed with 3-drug antiemetic regimen (ondansetron + aprepitant + dexamethasone), highly effective in 2026 |
| Kidney toxicity (nephrotoxicity) | Common | Moderate | Aggressive pre- and post-hydration; avoid NSAIDs; monitor creatinine |
| Peripheral neuropathy | Common | Mild–Moderate | Dose adjustments; calcium/magnesium infusions; switch to carboplatin if progressive |
| Hearing loss (ototoxicity) | Occasional | Mild–Moderate | Baseline and periodic audiograms; dose reduction if detected |
| Electrolyte imbalances | Common | Mild | Magnesium, potassium, calcium monitoring and supplementation |
| Anemia | Common | Mild–Moderate | Iron supplementation; erythropoietin-stimulating agents if severe |
| Fatigue | Very common | Moderate | Activity pacing; treat underlying anemia |
Carboplatin Side Effects (Comparison to Cisplatin)
Carboplatin generally produces:
- Less nausea, significantly more tolerable for most patients
- Less kidney toxicity, major advantage for patients with compromised renal function
- Less neuropathy, less peripheral nerve damage
- More bone marrow suppression, lower platelet counts (thrombocytopenia) more common; monitor carefully
- Equivalent efficacy, same survival outcomes as cisplatin when combined with pemetrexed
Managing Side Effects in 2026, What’s Changed
Side effect management has improved dramatically over the past decade:
Anti-nausea: The combination of 5-HT3 antagonists (ondansetron), NK1 antagonists (aprepitant), and dexamethasone now controls chemotherapy-induced nausea in 70–80% of patients. This triple combination has transformed the cisplatin experience, what was once debilitating nausea is now manageable for most patients.
Fatigue: Recognized as the most impactful side effect for quality of life, cancer-related fatigue is now addressed proactively with:
- Graded exercise therapy (counterintuitively, low-intensity exercise during chemo reduces fatigue)
- Sleep hygiene protocols
- Treatment of contributing factors (anemia, depression, thyroid dysfunction)
- Scheduled rest periods and activity pacing
Neutropenia (low white count): G-CSF injections (filgrastim/Neupogen or pegfilgrastim/Neulasta) given after chemo can prevent dangerous drops in white blood cell counts. Your oncologist determines if prophylactic G-CSF is appropriate for your protocol.
Mesothelioma Chemotherapy Survival Rate, 2026 Data
| Regimen | Median Overall Survival | 1-Year Survival | 2-Year Survival |
|---|---|---|---|
| Pemetrexed + Cisplatin | 12.1 months | 56.1% | 23.0% |
| Pemetrexed + Carboplatin | ~11–13 months | ~54% | ~22% |
| Pemetrexed + Cisplatin + Bevacizumab | 18.8 months | ~70% | ~40% |
| Pembrolizumab + Pemetrexed + Platinum | 17.3 months | ~70% | ~38% |
| Nivolumab + Ipilimumab (immunotherapy) | 18.1 months | ~73% | 41% |
| Chemo + Surgery (multimodal) | 20–31 months | ~75% | ~50–65% |
Sources: Vogelzang et al., JCO; MAPS trial, Lancet Oncology; KEYNOTE-483; CheckMate 743; published surgical series 2026
Key insight: Chemotherapy as a standalone treatment achieves median survival of 12–19 months. Combined with surgery and/or radiation in multimodal protocols, survival extends significantly. Combined with immunotherapy (bevacizumab or pembrolizumab), it achieves meaningful improvements over standard chemotherapy alone.
Chemotherapy vs. Immunotherapy, How to Choose
The 2024 ASCO guideline update formalized the treatment decision based on cell type:
| Cell Type | Preferred First-Line Option |
|---|---|
| Non-epithelioid (sarcomatoid/biphasic) | Immunotherapy, nivolumab + ipilimumab |
| Epithelioid | Choice between: chemotherapy alone, immunotherapy alone, or chemoimmunotherapy (pembrolizumab + chemo) |
| Either type, unresectable | Discuss all options with mesothelioma oncologist |
For epithelioid mesothelioma specifically: The decision between chemotherapy-first, immunotherapy-first, or a combination is now genuinely personalized, and depends on:
- The patient’s tolerance for different side effect profiles (chemo vs. immunotherapy have distinct toxicity patterns)
- Whether the patient is a potential surgical candidate (some surgeons prefer chemotherapy before surgery to test tumor responsiveness)
- Comorbidities that may favor one approach
- Patient preference after informed discussion
This is a conversation to have with a mesothelioma oncologist, not a general oncologist.
The Cost of Mesothelioma Chemotherapy
Chemotherapy for mesothelioma is expensive, and understanding costs helps families plan and pursue compensation.
| Treatment | Approximate Cost Per Cycle | Full Course (6 cycles) |
|---|---|---|
| Pemetrexed (Alimta) | $8,000–$12,000 | $48,000–$72,000 |
| Cisplatin | $500–$1,500 | $3,000–$9,000 |
| Carboplatin | $300–$800 | $1,800–$4,800 |
| Bevacizumab (Avastin) | $5,000–$8,000 | $30,000–$48,000 |
| Pembrolizumab (Keytruda) | $10,000–$15,000 | $60,000–$90,000 |
| Infusion center fees | $1,000–$3,000 | $6,000–$18,000 |
| Total (pemetrexed + cisplatin, full course) | , | ~$60,000–$90,000 |
| Total (pembro + pemetrexed + platinum) | , | ~$130,000–$200,000 |
How Chemotherapy Costs Are Covered
Medicare: Covers FDA-approved chemotherapy under Part B (physician-administered) and Part D (oral drugs). Pemetrexed + cisplatin/carboplatin and pembrolizumab + chemotherapy are all covered under current FDA approvals.
Commercial Insurance: All FDA-approved first-line regimens are covered by major commercial insurers.
Manufacturer Assistance Programs:
- Eli Lilly (pemetrexed/Alimta): Lilly Cares Foundation patient assistance program for uninsured or underinsured patients
- Merck (pembrolizumab/Keytruda): Merck Patient Assistance Program for eligible patients
- Roche (bevacizumab/Avastin): Genentech BioOncology Patient Assistance Program
Legal Compensation: Every dollar of mesothelioma chemotherapy costs is fully compensable in your asbestos lawsuit. Your attorney documents all treatment expenses as economic damages, recovering them from the manufacturers responsible for your exposure.
Chemotherapy and Your Legal Rights
Chemotherapy is expensive, physically demanding, and deeply disruptive to daily life and work. Every aspect of that burden was caused by companies that manufactured and sold asbestos products while knowingly concealing the dangers.
Your legal compensation claim covers:
- All chemotherapy drug costs, documented economic damages
- Infusion center fees, documented medical expenses
- Lost wages during treatment cycles when you couldn’t work
- Travel costs to and from infusion appointments
- Caregiver costs for help needed during treatment
- Pain and suffering, the physical impact of chemotherapy side effects
- Future treatment costs, anticipated ongoing medical expenses
And it runs completely parallel to your treatment. While you’re receiving pemetrexed infusions, your attorney is filing your lawsuit and trust fund claims simultaneously, at zero upfront cost to you.
| Compensation Source | Amount |
|---|---|
| Lawsuit Settlement | $1M – $1.4M average |
| Trial Verdict | $5M – $11.4M average |
| Asbestos Trust Funds | $300K – $400K combined |
| VA Benefits (veterans) | $4,158+/month |
| Combined Total | $1.5M – $2M+ |
Frequently Asked Questions
What chemotherapy is used for mesothelioma in 2026?
The standard first-line regimen is pemetrexed (Alimta) combined with cisplatin, FDA-approved since 2004. Carboplatin is substituted for patients who can’t tolerate cisplatin. A newer option is pembrolizumab (Keytruda) combined with pemetrexed and a platinum drug, FDA approved in 2025 based on the KEYNOTE-483 trial. Bevacizumab can be added for eligible patients.
How effective is chemotherapy for mesothelioma?
Pemetrexed + cisplatin achieves a 41% objective response rate and 12.1-month median survival. Adding bevacizumab improves median survival to 18.8 months. Adding pembrolizumab improves median survival to 17.3 months and increases response rates to 57%. Combined with surgery in multimodal protocols, survival extends to 20–31 months.
What are the worst side effects of mesothelioma chemotherapy?
The most commonly reported serious side effects are fatigue, nausea/vomiting (particularly with cisplatin), reduced blood counts (increasing infection risk), and neuropathy (numbness/tingling). Modern anti-nausea medications control nausea in 70–80% of patients. Folic acid and vitamin B12 supplementation significantly reduce pemetrexed-related toxicity.
How long does mesothelioma chemotherapy last?
A standard course is 4–6 cycles, each 21 days apart, totaling 12–18 weeks of treatment. Some patients receive additional maintenance cycles if they respond well. Neoadjuvant chemotherapy (before surgery) typically involves 3–4 cycles.
Can I work during chemotherapy?
Many patients continue working part-time during pemetrexed + carboplatin treatment. The days immediately following each infusion (days 2–5) typically involve the most fatigue and nausea. Cisplatin protocols tend to be more disruptive. Individual tolerance varies significantly, discuss your work schedule with your oncologist.
Does chemotherapy affect my legal case?
No, your legal rights are completely unaffected by your treatment decisions. Your chemotherapy costs are actually additional compensable damages in your asbestos lawsuit. Your attorney pursues your claim while you focus on treatment.
Is chemotherapy still used if I have immunotherapy?
Yes, in many protocols. Pembrolizumab is now FDA-approved in combination with pemetrexed + platinum, using both together. Patients who fail immunotherapy may proceed to chemotherapy. Patients receiving chemotherapy after surgery often add immunotherapy afterward. The two approaches are increasingly used together rather than sequentially.
Your Treatment Is the Priority. Your Legal Rights Run Alongside It.
Chemotherapy for mesothelioma is demanding. It takes time, causes side effects, and disrupts everything. But it can also meaningfully extend your life, and in combination with surgery and immunotherapy, it has helped patients achieve survival outcomes that would have seemed impossible a decade ago.
Your medical team manages your treatment. The firms connected through this site manage your legal case, simultaneously, at zero upfront cost. You focus on getting through treatment. They focus on recovering the compensation your family deserves from the companies that caused this.
👉 Mesothelioma Surgery Options 2026: EPP vs P/D, Which Is Right for You?
About the Author
Brian Watson is a senior research editor at MesotheliomaLegalHelp.info with over eight years of experience covering legal and medical topics for digital health publications. He specializes in asbestos litigation, occupational disease, veterans’ benefits, and mesothelioma treatment research. Brian’s work is reviewed against current medical literature, legal precedent, and publicly available case data before publication. He holds a degree in journalism with a concentration in health and science writing.